class 12 chemistry question paper 2025 with solution

















56/1/1 – Set
MARKING SCHEME
Sr. SECONDARY SCHOOL EXAMINATION, 2020
Subject: CHEMISTRY
Expected Answer / Value Points
SECTION – A
1 -Racemic Mixture
2- Polarimeter
3- Pent-2-ene / CH3CH=CHCH2CH3
4- Antiseptic
5-CH3I + C6H5OH
6- A
7- Zn
8-No
9- CH2=CH-Cl
10- Branched hydrocarbon part 1
11- B
12- D
13- C
14- C
15- A
16- iii
17- ii
18- i
19- ii
20- i
SECTION – B
Ans.21-(a) The drugs which are used to control stress / anxiety / tension / mild or severe mental diseases
(b) The drugs which are used to kill or to prevent the growth of micro-organism, applied externally on living tissues.
OR
Ans.21-Soap molecules form micelle around the oil droplet or dirt in such a way that hydrophobic part interacts with the oil droplet and hydrophilic part projects out. Micelles can be washed away on rinsing with water. Thus soap helps in emulsification and washing away of oil and fats.
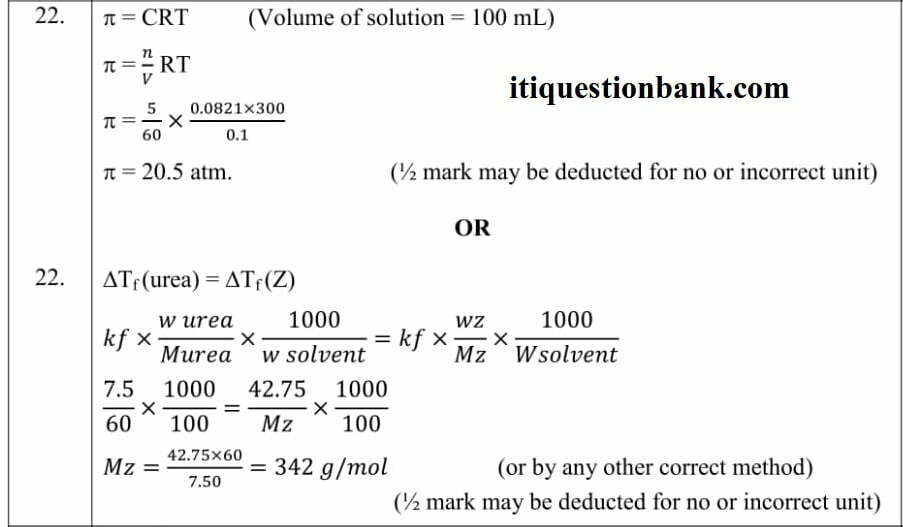
Ans.23(a) 1st order
(b) No, due to exponential relation / the curve never touches the x-axis.
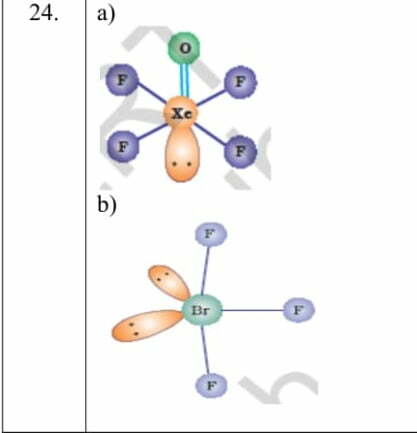
Ans.24 –
Ans.25-(a) K2[Zn(OH)4]
(b) [Pt(NH3)6]Cl4
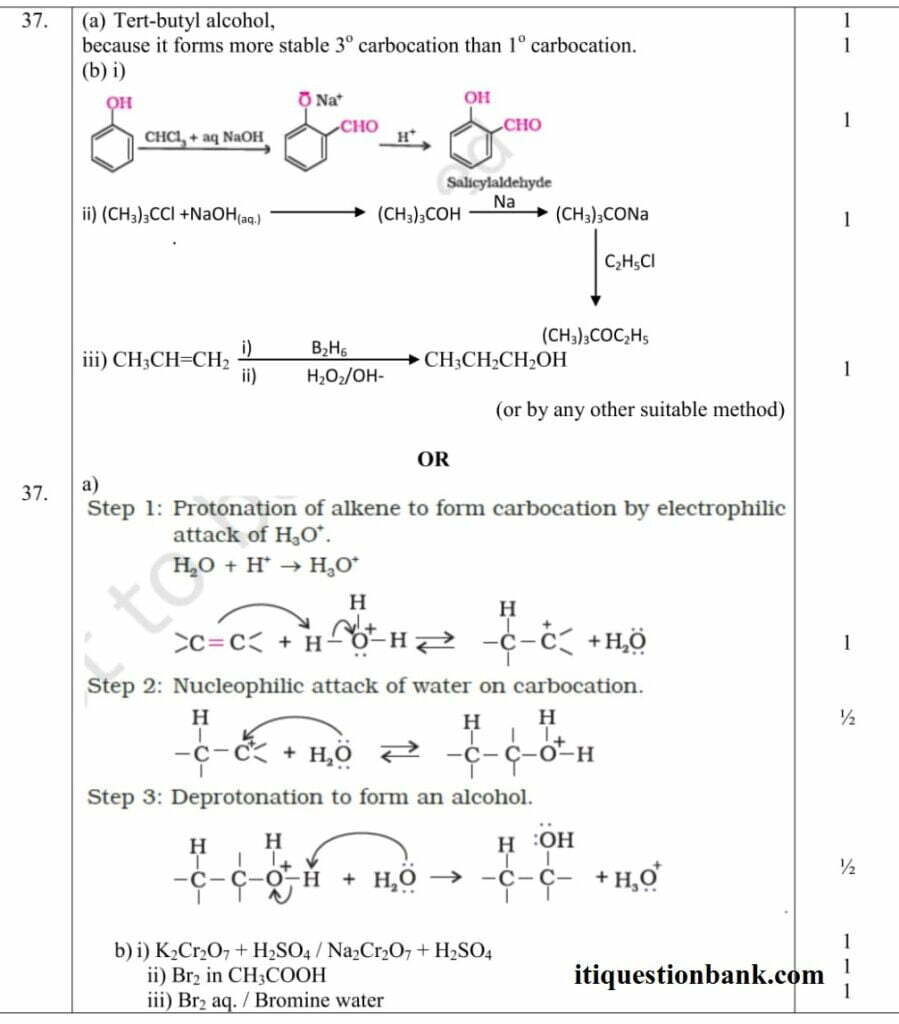
Ans.26 (a) (CH3)3C-OH / tertiary butyl alcohol is formed.
b) C6H5COCH3 / acetophenone is formed
Ans.27-(a) C6H5OH + HCHO , Phenol + formaldehyde
(b) CH2= C(Cl) – CH=CH2, Chloroprene
SECTION – C
Ans.28-(a) (A) → CH3CONH2 (B) → CH3NH2
(b) (A)→ C6H5NH2 (B) → C6H5N2Cl
(c) (A) → C6H5CN (B) → C6H5COOH
OR
Ans.28-(a) (i) Add Ice cold (NaNO2 + HCl) followed by phenol or β-Naphthol to both the compounds.
Aniline forms orange red dye while ethylamine doesn’t.
(ii) Add CHCl3 and KOH (alc.) to both the compounds.
Aniline gives foul smelling isocyanide while N-Methylaniline doesn’t.
(or any other suitable chemical test)
b) Butanol > Butanmine > Butane
Ans.29-(a) Because the – CHO group in glucose is involved in hemiacetal formation and thus is not free / due to cyclic structure of glucose -CHO group is not free.
(b) Because the hydrogen bonds are formed between specific pairs of bases.
(c) Starch is a polymer of D – glucose while cellulose is a polymer of E – glucose
Ans.30-(a) Because sulphur readily gets oxidized itself to more stable +6 state.
(b) Because of absence of d-orbital in Fluorine.
(c) Because size increases from Helium to Radon. / dispersion or van der Waal forces increase from Helium to Radon
OR
Ans.30 – (a) MnO2+ 4HCl → MnCl2 + Cl2 + 2H2O
(b) XeF6 + KF → K+[XeF7]–
(c) 4I–(aq.) + 4H+(aq.) + O2(g) → 2I2(s) + 2H2O(l)
Ans.31-(a) NaCN act as a depressant.
(b) SiO2 act as a flux. / used to remove FeO as slag
(c) I2 is used to convert Ti into volatile compound (TiI4)
Ans.32-
| Pystsorption | Chemisorption |
| it arise because of van der Waals force | it is cause by chemical bond formation |
| it is not specific in nature | It is highly specific in nature |
| it is reversible in nature. | It is irreversible |
Ans.33-(a) Decreases.
(b) Increases
(c) Increases
Ans.34 – △Tf = Kf m
1.5=(3.9xWBx1000)/176×75
Mass of ascorbic acid = 5.08g
SECTION – D
Ans.35 – (a) E0 cell = Eoc – EoA
= 0.34 – (-0.76)
= 1.10V
△Go = -nFEo
= -2 x 1.10 x 96500
= -212300 J/mol or -212.3 kJ/mol
(b) (i) Pollution free
(ii) High efficiency.
OR
Ans.35-(a) (i) Silver wire at 30o C because as temperature decreases, resistance decreases so conduction increases.
(ii) 0.1 M CH3COOH, because on dilution degree of ionization increases hence conduction increases.
(iii)KCl solution at 50o C, because at high temperature mobility of ions increases and hence conductance increases
| (b) Electrochemical | Electrolytic |
| Anode -ve Cathode +ve | Anode +ve Cathode -ve |
| Convert chemical Energy to electrical energy | Convert electrical Energy to chemical energy |
Ans.36-
(a) (i) Cu+1(3d10) compounds are white because of absence of unpaired
electrons while Cu+2 (3d9) compounds are coloured due to unpaired e- / shows d- d transition.
(ii) chromate (CrO42-) changes to dichromate (Cr2O72-) ion in acidic medium.
(iii)due to completely filled d-orbitals in their ground state as well as in oxidized state.
(b) Co = [Ar]4s2 3d7 , Co+2 =[Ar] 3d7
μ=√n(n+2)
=√3(3+2)
=√15 =3.92B.M
OR
Ans.36-
| (a) Lanthanoids | Actinoids |
| most of them are not radioactive | All are radioactive |
| don’t show a wide range of oxidation state | Show a wide range of oxidation states |
| Most of their ions are colourless | Most of their ions are coloured |
(b) (i) Sc+3, because of absence of unpaired electron.
(ii) Cr, because of presence of strong intermetallic bonding than Cu
ये भी पढ़े …….
